The total quantity of veterinary antibiotics sold in Ireland during 2019 was 88.8t, considerably lower than recent years and comparable with a figure of 88.3t in 2009.
The data, collected by the Health Products Regulatory Authority (HPRA) during 2019, details the sales of all veterinary antibiotics marketed in Ireland.
Reductions in several classes were noted, most importantly the sales of highest-priority critically important antibiotics.
Changing trends
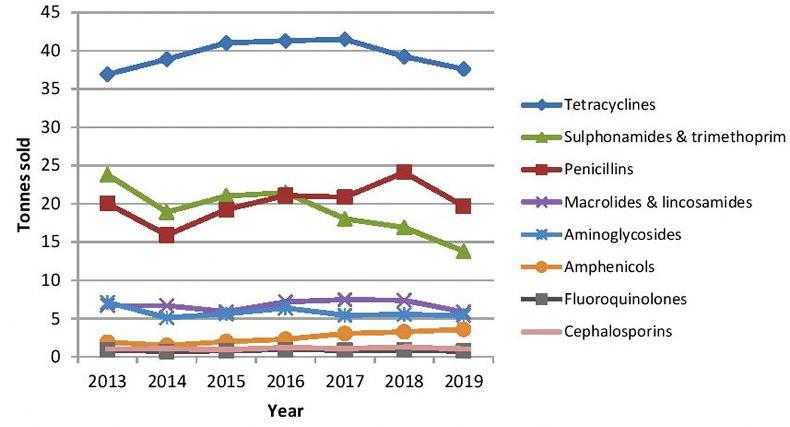
Sales (tonnes) of veterinary antibiotics for the years 2013 to 2019.
As detailed in the graph, the sales of the highest-priority critically important antibiotics cephalosporins, fluoroquinolones and macrolides fell in 2019.
A minor shift from injectables and oral remedies back to premixes was also observed. Pharmaceutical breakdown of veterinary antibiotics sold in 2019 included:
Premix - 37.4%.Oral remedy - 34.1%.Injectable - 25.5%.Intramammary dry -1.6%.Tablet - 1.0%.Intramammary milking - 0.5%.Other - less than 0.05%.National initiatives
HPRA has described the findings as "encouraging" and said they may be the result of national initiatives by a number of stakeholders to encourage prudent use of antimicrobials.
“While the reduction in sales of antibiotics in Ireland during 2019 is encouraging and very welcome, it should be noted that the sales of antibiotics used in animals throughout the EU fell by more than 34% between 2011 and 2018.”
New Veterinary Medicines Regulation, which comes into force in January 2022, requires that data on antibiotic sales be complemented using data on actual usage in animals.
The requirement to collect use data, additional to sales data, is expected to commence in 2023 and proceed on a phased basis to encompass all species by 2029.
This new information will hopefully allow a better understanding of how veterinary antibiotics are used in practice and will help pinpoint whether further regulatory measures are needed to ensure responsible use.
Practical guidance
The IFA has said that the real solutions to AMR are to be found outside the farm gate and must be addressed to mirror the work of farmers.
“Clearly the sector specific prudent use guidelines for the responsible prescribing and use of antibiotics on farms are gaining traction and informing decision making.
“This approach proves how progress can be made by providing practical advice and guidance without the need for a blunt legislative approach. This should be taken on board in the broader discussion on implementation of new regulation.”
Read more
No wiggle room for Ireland on tighter animal medicine rules
Department to tackle vets who over-prescribe medicines
Antibiotic use down 22% in Northern Ireland
The total quantity of veterinary antibiotics sold in Ireland during 2019 was 88.8t, considerably lower than recent years and comparable with a figure of 88.3t in 2009.
The data, collected by the Health Products Regulatory Authority (HPRA) during 2019, details the sales of all veterinary antibiotics marketed in Ireland.
Reductions in several classes were noted, most importantly the sales of highest-priority critically important antibiotics.
Changing trends

Sales (tonnes) of veterinary antibiotics for the years 2013 to 2019.
As detailed in the graph, the sales of the highest-priority critically important antibiotics cephalosporins, fluoroquinolones and macrolides fell in 2019.
A minor shift from injectables and oral remedies back to premixes was also observed. Pharmaceutical breakdown of veterinary antibiotics sold in 2019 included:
Premix - 37.4%.Oral remedy - 34.1%.Injectable - 25.5%.Intramammary dry -1.6%.Tablet - 1.0%.Intramammary milking - 0.5%.Other - less than 0.05%.National initiatives
HPRA has described the findings as "encouraging" and said they may be the result of national initiatives by a number of stakeholders to encourage prudent use of antimicrobials.
“While the reduction in sales of antibiotics in Ireland during 2019 is encouraging and very welcome, it should be noted that the sales of antibiotics used in animals throughout the EU fell by more than 34% between 2011 and 2018.”
New Veterinary Medicines Regulation, which comes into force in January 2022, requires that data on antibiotic sales be complemented using data on actual usage in animals.
The requirement to collect use data, additional to sales data, is expected to commence in 2023 and proceed on a phased basis to encompass all species by 2029.
This new information will hopefully allow a better understanding of how veterinary antibiotics are used in practice and will help pinpoint whether further regulatory measures are needed to ensure responsible use.
Practical guidance
The IFA has said that the real solutions to AMR are to be found outside the farm gate and must be addressed to mirror the work of farmers.
“Clearly the sector specific prudent use guidelines for the responsible prescribing and use of antibiotics on farms are gaining traction and informing decision making.
“This approach proves how progress can be made by providing practical advice and guidance without the need for a blunt legislative approach. This should be taken on board in the broader discussion on implementation of new regulation.”
Read more
No wiggle room for Ireland on tighter animal medicine rules
Department to tackle vets who over-prescribe medicines
Antibiotic use down 22% in Northern Ireland










SHARING OPTIONS